From BioNet-Asia
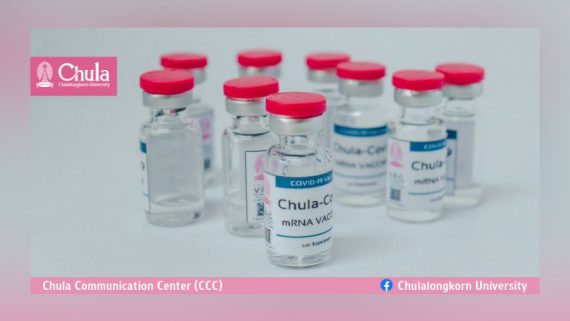
ChulaCov19 BNA159 is a COVID–19 mRNA vaccine developed by Chulalongkorn University and produced by BioNet
- ChulaCov19 mRNA vaccine was found safe and immunogenic for primary immunization in clinical trials in Thailand.
- The Australian trial will evaluate the safety and immunogenicity of a booster dose of ChulaCov19 BNA159 mRNA vaccine in healthy volunteers.
- The collaborators of the trial are Chulalongkorn University, BioNet and Technovalia.
Melbourne, 15th November 2022 – Technovalia, Chulalongkorn University and BioNet today announced that Human Research Ethics Committee (HREC) in Australia approved the phase 2 trial of ChulaCov19 BNA159 mRNA vaccine as a booster dose in adults.
Ethics approval is granted to commence a phase 2 trial after sharing satisfactory results of clinical safety and immunogenicity data of prior phase 1 and 2 studies of ChulaCov19 mRNA vaccine.
Mr. Laurent Dapremont, Chief Executive Officer of Technovalia said: “We are pleased to have received ethics approval for testing the ChulaCov19 mRNA BNA159 vaccine. We have been leading the development of genetically designed innovative vaccines in Australia, starting with clinical trials evaluating a recombinant pertussis vaccine, two DNA COVID vaccines and now a mRNA COVID vaccine.”
The clinical trial will assess the safety and immunogenicity of one single booster dose of ChulaCov19 BNA159 mRNA vaccine in healthy volunteers having previously received vaccination with an approved COVID-19 vaccine. The trial includes several sites in Australia and will commence in November.
Assoc. Prof. Chanchai Sittipunt, MD, Dean of Faculty of Medicine, Chulalongkorn University added: “Our university aims at developing approaches to move society forward. Developed in Thailand, Chulacov19 mRNA vaccine has already shown good safety data and very promising immunogenicity data in Thai phase 1 and phase 2 trials. We are glad to continue its development as a booster vaccine and to sponsor this clinical study in Australia. We believe mRNA vaccines will play an important role to combat current and future pandemics and infectious disease threats.”
ChulaCov19 mRNA vaccine was developed by Professor Kiat Ruxrungtham and his team of Chulalongkorn University Vaccine Research Center (ChulaVRC), Thailand, in collaboration with Professor Drew Weissman of the University of Pennsylvania, USA. the first ChulaCov19 vaccine lot was manufactured in the U.S. and was evaluated in a phase 1 safety trial in Thailand. It was then evaluated in a comparative phase 2 safety and immunogenicity trial in Thai adults and elderly participants who received two primary doses of vaccine. The ChulaCov19 vaccine was compared to an mRNA vaccine licensed in Thailand. The new multi-centre trial in Australia, will evaluate the boosting immunity induced by one dose of ChulaCov19 BNA159 vaccine, produced in Thailand, in adults aged 18 – 64 years.
Dr. Pham Hong Thai, Chief Executive Officer of BioNet, added: “We are very pleased to collaborate with Chulalongkorn University to produce mRNA vaccines against SARS–CoV–2 and to continue our development partnership with Technovalia. We have been collaborating on several vaccines of which three are in clinical trial stage in Australia. This is a key milestone for all our colleagues who worked relentlessly towards the success of this mRNA vaccine project.”
BioNet has established an end-to-end manufacturing platform to produce mRNA vaccines from cell bank to encapsulation in record time. ChulaCov19 BNA159 mRNA vaccine is the fruit of a collaboration between Chulalongkorn University and BioNet. The mRNA technology platform enables the rapid development of new vaccines in 100 days from research to use in human trial. A second-generation mRNA vaccine against Omicron variant and the ancestral strain of SARS-CoV-2 virus is also in development.
About Chulalongkorn University
Chulalongkorn University aims to become a model institute of education, setting the standard as a university of innovations for society and focusing on three social development core principles: preparing future leaders, developing impactful research and innovation, and advocating social sustainability. The university has 20 faculties, 23 colleges and research institutes, more than 3,000 full-time faculty members and over 37,000 students. Recently, Chulalongkorn University was ranked Asia’s No.1 for Global Impact by Times Higher Education (THE) Impact Ranking 2021, which is the global performance scale that assesses universities using the United Nations’ 17 Sustainable Development Goals (SDGs). Chulalongkorn University earned the scores of SDG 3 for Good Health and Well-being and SDG 9 for Industry, Innovation and Infrastructure.
About BioNet
BioNet is a biotech organization sharing expertise and innovation to secure rapid access to vaccines. Located in France and Thailand, BioNet creates genetically designed vaccines and produces Pertagen, the world’s only recombinant pertussis-only vaccine containing genetically inactivated pertussis toxin. BioNet has also established technology platforms to produce DNA and mRNA vaccines with 10 projects in preclinical and clinical studies. BioNet is advancing global research in collaboration with eminent advisors and organizations (The Pasteur Institute, The Bill and Melinda Gates Foundation, PATH, CEPI and IVI). BioNet has developed bio-clusters fostering vaccine self-reliance around the world and for 20 years, BioNet has driven a unique manufacturers alliance deploying 10 billion doses of polio vaccines worldwide.
About Technovalia
Melbourne-based Technovalia is a privately-owned Australian biotech company dedicated to the research and development of innovative vaccines. In partnership with several academic organisations and international companies, Technovalia is investing in the development of new technology platforms that have the potential to significantly improve protection against several infectious diseasesby producing safer, more stable, and more cost-effective vaccines. Technovalia is developing BioNet’s recombinant acellular pertussis-only vaccine Pertagen® in Australia and needle-free COVID-19 DNA-based vaccine Covigen.
For further information, please contact:
Ms Michelle Tat
Marketing Communications
E-mail: info@technovalia.com
Amid a global shortage of vaccines and a new wave of COVID-19 infections, some countries in Southeast Asia are rushing to develop their own coronavirus shots.
COVID19 Vaccine Tracker – Chulalongkorn University: ChulaCov19
The research team behind the development of a local mRNA Covid-19 vaccine on Monday asked the government to help finance the project
The Phase 1 trial of Thailand’s homegrown mRNA COVID-19 vaccine ChulaCOV-19, suggests the candidate vaccine can help the body elicit
The mRNA vaccine, currently being developed by the Vaccine Research Centre of the Faculty of Medicine of Chulalongkorn University, can stimulate the immune system to produce killer T-cells
The Faculty of Medicine at Bangkok’s Chulalongkorn University has put out a call for healthy volunteers to take part in trials of its Covid-19 mRNA vaccine.
TriLink enables Chula VRC to take its Covid-19 vaccine into a First in Human (FIH) phase 1 clinical trial in Thailand with mRNA
With Covid-19 battering Thailand and the third wave bringing the Sinovac-resistant Delta variant to the forefront of the pandemic,
Prof Kiat Ruxrungtham from the Center of Excellence in Vaccine Research Center and Development. Photo courtesy of Chulalongkorn Hospital
The first phase of clinical trial on volunteers who have passed the screening process and deemed to be in good health
Chulalongkorn University’s mRNA Covid-19 vaccine, ChulaCov19, began its first round of human trials
ChulaCov19 designer Kiat Ruxrungtham talks about his aim to make Thailand an mRNA vaccine hub